Aditif Dispersing Agent
Perannya di Dalam Sistem Pigmen
SURFACE SCIENCEPUBLIC
Arif Sarwono
11 min baca
There is a component in your formulation that weighs less than 2% of the total batch, costs a fraction of your pigment spend, and yet - get it wrong - and nothing else you do will save the color. Viscosity climbs. Gloss drops. Strength is lost. The grind never finishes. And the worst part: it all looks fine in the drum, until it doesn't.
That component is the dispersing agent. And most formulators know that it works. Fewer understand why - at the level where you can make a confident decision when the standard recommendation fails you.
This article is for the latter group.
The Problem You Are Actually Solving
Before we talk about dispersants, we need to be precise about the problem.
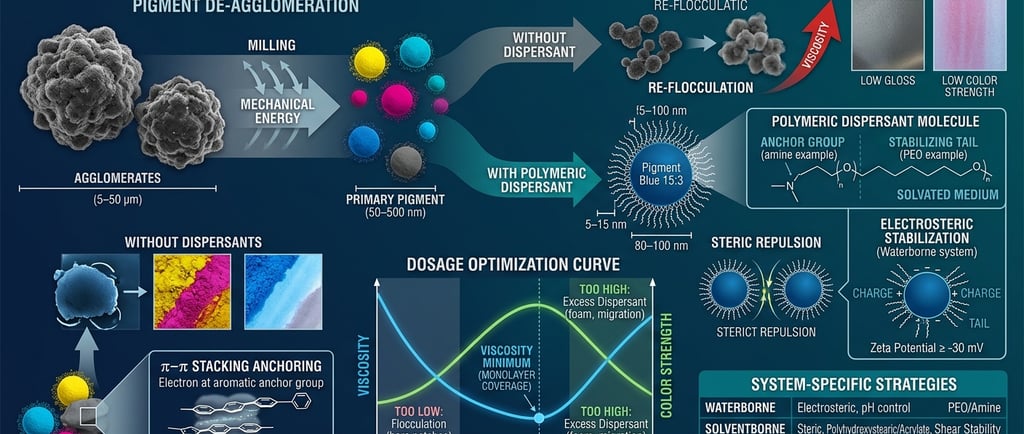
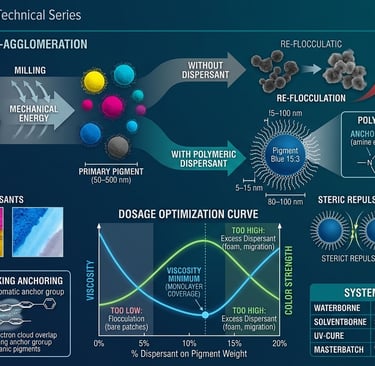
When you receive a pigment - organic, inorganic, carbon black, effect pigment - what you are receiving is not a collection of individual primary particles. You are receiving agglomerates: loosely bound clusters of primary particles held together by Van der Waals attractive forces.[1,7] These clusters can range from 5 µm to 50 µm in diameter. Your primary particles - the actual chromophores doing optical work - are sitting somewhere between 50 nm and 500 nm, buried inside.[3]
The job of milling is to break those agglomerates back down to primary particle level. The job of the dispersing agent is to make sure they stay broken down.[1,2]
This is the distinction that matters. Milling alone, without dispersant, will break agglomerates. But the moment the mechanical energy stops, the freshly exposed surfaces — now carrying enormous surface energy - will find each other again. You get re-flocculation: a soft, reversible clumping that looks different from the original agglomerate but produces the same result. Color strength is lost. Viscosity rises. Transparency collapses.[1,2]
The dispersing agent's role is to occupy those freshly exposed surfaces before re-flocculation can happen, and then hold particles apart permanently through a repulsive force that exceeds the Van der Waals attraction pulling them together.[1,4,7]
The Molecule Doing the Work
A dispersing agent is not a simple surfactant - though the distinction is often blurred in supplier literature. Understanding the structural difference matters because it explains performance differences you will observe in the lab.[5]
A conventional surfactant has one hydrophilic head and one hydrophobic tail. It is designed for liquid-liquid interfaces (emulsification) or air-liquid interfaces (foaming/wetting). Its affinity for a solid pigment surface is incidental, not engineered. It adsorbs weakly, desorbs easily under shear or temperature change, and provides minimal steric protection because the tail is short.[5,6]
A polymeric dispersant - what you are using when you reach for your typical dispersant such as Solsperse, Disperbyk, TEGO Dispers, or similar - is architecturally different:
The anchor group is a chemically specific moiety engineered to bind tightly to a particular pigment surface chemistry. Amine groups (–NH₂, –NH–) anchor on acidic pigment surfaces. Carboxylate, phosphate, and sulfonate groups anchor on basic surfaces.[5,6] Aromatic anchor groups - or pigment synergists - anchor on organic pigment crystal faces through π–π stacking interactions.[10,11] Unlike simple surfactants that physisorb weakly and desorb readily under shear, polymeric dispersants with multiple anchor points achieve strong, effectively irreversible adsorption - the probability of all anchor points simultaneously detaching approaches zero.[4]
The stabilizing tail is a polymer chain extending into the medium. Its only job is to create a physical barrier between particles. When two coated particles approach each other, the tails compress. Compressed polymer chains lose entropy - thermodynamics pushes the particles apart. This is steric repulsion, and it is the primary stabilization mechanism in solventborne systems.[4,6]
For the tail to work, it must be solvated by the medium - the solvent or resin must be a good solvent for the tail polymer. A tail that collapses against the particle surface provides no protection at all. This is why you cannot use an aliphatic polyester-tailed dispersant in a highly polar waterborne system, and why a PEO-tailed dispersant designed for water may not function correctly in a mineral-spirit-based alkyd.[4,5,6]
In waterborne systems, a second mechanism operates in parallel: electrostatic repulsion. Ionic groups on the dispersant (or on the pigment surface directly) impart a surface charge - measurable as zeta potential. When two particles carry the same sign charge, they repel. The threshold for stability is generally accepted as ≥ ±30 mV zeta potential.[7] Below this, the electrostatic barrier is insufficient and flocculation occurs. The combination of both mechanisms - charge and steric - is called electrosteric stabilization, and it is why well-formulated waterborne systems can achieve stability with lower dispersant loadings than their solventborne equivalents.[1,7]
The Scale Relationship You Need to Visualize
Here is a mental model that resolves a lot of confusion.
A primary pigment particle - say, a Pigment Blue 15:3 particle at its primary size - is roughly 80–100 nm in diameter.[3] A polymeric dispersant molecule is 5–15 nm in total length. The ratio is approximately 10–20:1.
This means one pigment particle is not coated by one dispersant molecule. It is coated by hundreds to thousands of dispersant molecules - anchor heads gripping the surface, polymer tails radiating outward into the medium like the fur of an animal. The resin is not a shell around this coated particle; it is the continuous ocean in which these coated particles swim.[1,6]
The implication for dosage is direct and calculable. A single gram of Pigment Yellow 83, with a specific surface area (SSA) of approximately 40 m²/g, [3,9] carries 40 square meters of surface that needs to be covered. A polymeric dispersant with a molecular weight of around 4,500 g/mol occupies roughly 4–5 nm² per molecule on that surface. Working through the arithmetic:
Coverage needed: 40 m² × 10¹⁸ nm²/m² = 4 × 10¹⁹ nm²
Molecules needed: 4 × 10¹⁹ ÷ 4.5 nm²/molecule ≈ 8.9 × 10¹⁸ molecules
Mass of dispersant: 8.9 × 10¹⁸ ÷ 6.022 × 10²³ × 4500 g/mol ≈ 0.066 g
That is 6.6% of a 1g pigment sample - before any safety factor. Add a 1.5–2× multiplier to account for imperfect surface access, equilibrium adsorption dynamics, and newly exposed surfaces during milling, and you arrive at a practical dosage in the range of 10–15% for a fine organic pigment.
This is why a dispersant TDS for inorganic pigments shows 2–15%, and for organic pigments 5–35%. The range is not imprecision - it is the SSA range of the pigment class, encoded as a dosage window. Carbon black at 200 m²/g will push toward 30–35%. Ultramarine blue at 2 m²/g will sit at 2–3%.[9,13] The dispersant TDS cannot collapse that range further without knowing your specific pigment's SSA.[12]
This calculation assumes brush conformation - the ideal state where polymer tails extend fully into the medium. Non-ideal conformations (mushroom, flat) increase the molecular footprint, paradoxically reducing the theoretical dosage needed for surface coverage - but at the cost of steric protection.[4] A dispersant lying flat on a pigment surface covers more area per molecule but provides no repulsive barrier. This is why the theoretical dosage calculation is a starting point, not a final answer. The viscosity minimum method remains the only reliable way to locate the true optimum - because it captures coverage and conformation performance simultaneously in your actual system.[10,13]
Reading a TDS: What the Numbers Actually Mean
Take TEGO Dispers 656 as a working example. Its key data:[12]
Chemical description: Modified polyether with groups of high pigment affinity
Acid value: ~100 mg KOH/g
Active matter: 100%
Solubility: Soluble in water and ethanol; partly soluble in TPGDA and butylacetate; not soluble in mineral spirits
The acid value of 100 mg KOH/g is the most informative single number on that sheet.[6,12] The acid value confirms the presence of acidic anchor groups - engineered to bind strongly to the basic surfaces of inorganic pigments and effect pigments.[5,6] Acidic anchor groups bind strongly to basic surfaces. Inorganic pigments - TiO₂, ZnO, iron oxides, aluminum effect pigments - all carry basic surface character. This is why the TDS lists "outstanding stabilization of inorganic particles" as its first key benefit. The chemistry is not marketing language; it is a direct consequence of the acid-base match between anchor and surface.[5,6,12]
For organic pigments - which carry acidic or neutral surfaces - the acid anchor affinity is weaker. You are trying to adsorb an acidic group onto an acidic surface. It works, but inefficiently. You compensate with higher loading (5–35%) and accept lower color development efficiency compared to a dispersant with an amine anchor on the same organic pigment.[3,6,12]
The solubility data tells you about tail compatibility. Soluble in water and ethanol, the polyether tail is polar and hydrophilic, works in waterborne and alcohol-based systems. Partly soluble in butylacetate, marginal in medium-polarity solventborne. Insoluble in mineral spirits, do not use this in alkyd or oil-based inks.[12] A tail that is insoluble in your medium collapses. A collapsed tail provides zero steric protection. [4,5]The pigment flocculates, your viscosity climbs, and you spend weeks troubleshooting what looks like a grinding problem.
The "not suitable" rating for direct grind on the suitability matrix reflects this: without a resin to solvate the tail, stability is marginal in solventborne direct-grind applications.[12]
Where the Dosage Optimum Lives - and How to Find It
The correct dispersant dosage for your specific system is a single point, not a range. The range in the TDS brackets it - your job is to locate it experimentally.[10,13]
The method is a dosage-viscosity curve, sometimes called an adsorption isotherm in formulation practice. Fix your pigment loading. Fix your medium. Fix your grind time and intensity. Vary only the dispersant loading across a series of batches - say, 2%, 4%, 6%, 8%, 10%, 12% on pigment weight. Measure viscosity (at a consistent shear rate), grind fineness (Hegman gauge), and if possible, color strength via spectrophotometry.[13,14]
The viscosity curve will dip to a minimum. That minimum is where monolayer coverage is reached - every particle surface is coated, particles repel each other maximally, and the system flows freely.[10,13] To the left of the minimum, insufficient coverage means bare surface patches, particle-particle contact, and network viscosity. To the right of the minimum, excess dispersant accumulates as free material in the medium - it acts as a plasticizer, slightly raises viscosity, and in waterborne systems, contributes to foam and can reduce film hardness.[6,13]
The color strength curve typically peaks at or slightly before the viscosity minimum - another confirmation of the optimum. Maximum color strength equals maximum particle separation equals maximum optical path through individual particles. Flocculated particles optically behave as larger, less efficient absorbers.[9,14]
This optimum shifts with SSA variation between pigment batches from the same supplier - one reason why a formulation locked against one production lot may need dispersant adjustment against the next. This is not a formulation failure; it is BET surface area variation between manufacturing runs, which is real, documented, and routinely underestimated.[8,9]
The System-Specific Decisions
Different application systems require fundamentally different dispersant strategies — not just different products, but different mechanisms.
In waterborne inks and coatings, the dispersant must be compatible with pH shifts (your system may run pH 7.5-9.0), ionic strength from co-solvents and additives, and freeze-thaw cycling. Block copolymer dispersants with PEO tails and amine or carboxylate anchors are the standard.[1,5] Zeta potential measurement is your primary stability diagnostic - measure it at the pH of your formulation, not at neutral.[7]
In solventborne and high-solids systems, steric repulsion does all the work. Tail length and solvation quality are everything. Hyperdispersants - polyhydroxystearic acid-based for non-polar media, polyacrylate-based for polar solvents - are designed specifically for this environment.[1,5] The critical test is shear stability: measure viscosity before and after high-shear mixing. A system that flocculates under shear had inadequate steric protection to begin with.[4,6]
In masterbatch and plastic coloring, there is no liquid phase during dispersion. Everything happens in the melt - at 180–260°C, under the shear of a twin-screw extruder. Wax-based dispersants (polyethylene wax, polypropylene wax, fatty acid esters) work by lowering the interfacial tension between pigment and polymer melt, allowing the melt to wet and encapsulate each particle.[1,3] Organic pigments often require a pigment synergist - a modified version of the pigment itself, carrying anchor groups - added at 5–10% on pigment weight, working alongside the wax dispersant.[10,18] Without the synergist, the wax alone cannot prevent reagglomeration in the high-viscosity melt environment.
What You Are Missing If You Skip the Surface Chemistry
The single most common dispersant selection error is choosing by medium compatibility alone and ignoring anchor-surface match.[5,6] A dispersant that is fully compatible with your solvent or resin but has the wrong anchor group for your pigment will give you a partially stable system - one that passes on the day of manufacture, flocculates on storage, and fails the customer.
The check is straightforward:[5,6,3]
Pigment surface chemistry: Acidic (organic pigments, oxidized-grade-carbon black ) --> Anchor type needed: Amine, polyamine, imidazoline
Pigment surface chemistry: Basic (TiO₂, ZnO, iron oxides, CaCO₃) --> Anchor type needed: Phosphate ester, carboxylate, sulfonate
Pigment surface chemistry: Neutral / π-system (quinacridone, CuPc) --> Anchor type needed: Pigment synergist + amine or carboxylate
Pigment surface chemistry: Effect pigments (aluminum, mica) --> Anchor type needed: Modified polyether, silane, phosphate
Once anchor match is confirmed, then select for tail-medium compatibility. In that order. Every time.
The Number That Keeps Surprising Formulators
Return to the opening proposition: 1–2% by total formulation weight - sometimes less - is sufficient to stabilize the entire pigment fraction.
The reason is geometric. Dispersant is a surface treatment, not a matrix material. You need enough to coat the interface - a layer measured in nanometers - not enough to fill the space between particles. The resin fills that space. Dispersant covers the walls.[1,4]
For a 5% pigment loading in a final paint, with an inorganic pigment at 10 m²/g SSA, and a monolayer coverage requirement of ~0.25 mg/m², the arithmetic lands below 1% on total formulation weight. Knowing this makes you confident when a supplier recommends "2 - 15% on pigment" - you can calculate where in that range your system sits, rather than defaulting to the midpoint and hoping.[12,13]
Formulate from first principles. The data is on the TDS, the SSA is on the pigment specification sheet, and the calculation takes less than five minutes. The resulting formulation is more efficient, more stable, and better understood than anything arrived at by trial and addition.
Sources:
[1] Tadros, T.F. (2012). Dispersion of Powders in Liquids and Stabilization of Suspensions. Wiley-VCH, Weinheim.
[2] Patton, T.C. (1979). Paint Flow and Pigment Dispersion, 2nd ed. Wiley-Interscience, New York.
[3] Herbst, W., Hunger, K., Wilker, G., Ohleier, H., Winter, R. (2004). Industrial Organic Pigments: Production, Properties, Applications, 3rd ed. Wiley-VCH, Weinheim.
[4] Napper, D.H. (1983). Polymeric Stabilization of Colloidal Dispersions. Academic Press, London.
[5] Tadros, T.F. (2005). Applied Surfactants: Principles and Applications. Wiley-VCH, Weinheim.
[6] Bieleman, J. (ed.) (2000). Additives for Coatings. Wiley-VCH, Weinheim.
[7] Verwey, E.J.W., Overbeek, J.T.G. (1948). Theory of Stability of Lyophobic Colloids. Elsevier, Amsterdam.
[8] Brunauer, S., Emmett, P.H., Teller, E. (1938). "Adsorption of Gases in Multimolecular Layers." Journal of the American Chemical Society, 60(2), 309–319.
[9] ScienceDirect Topics / Elsevier. "Surface Pigment — BET Surface Area Measurement." https://www.sciencedirect.com/topics/engineering/surface-pigment
[10] Lawrence Industries / Münzing Chemie (2019). "Determining the Optimal Dispersant Loading for Pigments and Fillers." https://www.l-i.co.uk/knowledge-centre/determining-the-optimal-dispersant-loading-for-pigments-and-fillers/
[11] Chen, Z., et al. (2025). "Anion-π Type Polymeric Nanoparticle Dispersants for Enhancing the Dispersion Stability of Organic Pigments in Water." ACS Applied Materials & Interfaces. PMC11901968.
[12] Evonik Operations GmbH. TEGO® Dispers 656 Technical Data Sheet. Essen: Evonik (current edition).
[13] Uniqchem / Lawrence Industries (2019). "Dispersants for Steric Stabilisation." UL Prospector Knowledge Center. https://www.ulprospector.com/knowledge/5437/dispersants-for-steric-stabilisation/
[14] Günter, E., Zimmermann, F. (2011). "Optimizing Dispersant Levels for Waterborne Pigment Color Concentrates." PCI Magazine.
[15] Blackwell, C.S., et al. (1987). US Patent 4,673,705. "Radiation-curable coatings containing reactive pigment dispersants." USPTO.
[16] American Coatings Association (2019). "Improved Durability through Reactive Dispersant Technology." CoatingsTech. https://www.paint.org/coatingstech-magazine/articles/improved-durability-through-reactive-dispersant-technology/
[17] Faozi Nsib, et al. (2007). "Selection of dispersants for the dispersion of C.I. Pigment Violet 23 in organic medium." Dyes and Pigments, 73(1), 1–7. DOI: 10.1016/j.dyepig.2005.10.010.
[18] Kim, S., et al. (2019). "Improving nanoparticle dispersions of pigment and its application to a color filter: New phthalocyanine derivatives as synergist." Journal of Industrial and Engineering Chemistry, 69, 211–218.
Have a specific challenge in your production process?
All technical consultations and inquiries are treated as strictly confidential. Production details, formulation data, and business information you share are used only to serve your specific needs — never disclosed to third parties or supply chain partners without your consent. Reach out to Arif Sarwono at contact@mdm-technology.com
Hubungi kami kapan saja untuk mendapatkan kiat atau diskusi tentang teknik kimia atau aplikasi kimia.
Kontak KAMI
contact@mdm-technology.com
taramkt@gmail.com
+62811883915
+62818159065
© 2026. MDM Technology All rights reserved.